Diabetes treatments take many forms, but they all have the same purpose: regulating glucose in patients whose bodies cannot control glucose levels. Only within the last few years has technology evolved to permit continuous glucose monitoring outside hospitals. Continuous glucose monitoring helps doctors prescribe insulin therapies and dietary programs that are much better suited to the individual patient’s needs rather than a generic program that does not take a person’s unique daily glucose trends into account. Overall health in diabetic patients can be greatly improved when their blood glucose levels are maintained within a specified range.
Introduction
Diabetes is one of the most common maladies in the United States, affecting over 15.7 million people. Our nation spends over $100 billion annually in diabetes treatment and research, making this one of the most costly diseases in the world [1]. For years doctors have been treating diabetes using strict diets, exercise programs, and in severe cases, insulin injections. The success of each of these measures in preventing long-term health problems hinges on accurately measuring patients’ blood sugar (glucose) levels. Glucose sensing is an area of diabetic research that is booming with new developments and has the potential to drastically impact the way diabetes is treated. Various engineering companies have developed the technology for glucose sensing.
Diabetes
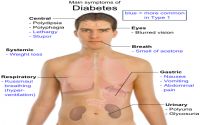
Diabetes mellitus is a chronic disease that affects the body’s ability to produce or respond to insulin, appearing with a variety of symptoms (see Fig. 1). Insulin is a hormone produced in the pancreas and it causes glucose (a simple sugar) to be absorbed into the blood stream and enter cells to be used for energy. People with diabetes cannot make enough insulin to meet their body’s needs. As a result, glucose builds up in the blood, overflows into the urine, and passes out of the body unused. Thus, the body loses an important source of fuel, even though the blood contains large amounts of glucose [2]. Our bodies, just like our cars, need fuel. You could not simply pour gasoline over the outside of your car and expect the vehicle to function. You must use a pump to place gas directly into the car’s tank where it will be broken down for power. Insulin works in our bodies like a gas pump works in a car. It directs glucose into the cells where it can be used for fuel.
The effects of diabetes are quite serious; without proper insulin levels the amount of glucose in the blood is irregular, which, in turn, disturbs many body functions. A safe glucose range is 75-120 mg of glucose/dl of blood. After eating, food is broken down into glucose molecules and is absorbed into the blood, raising glucose levels dramatically. For a normal, insulin-producing person the blood glucose level will fall quickly as glucose enters cells and is metabolized. If there is not sufficient insulin, glucose levels will remain very high because the cells will not be able to get needed fuel. This is a condition called hyperglycemia. For diabetics this is the primary problem and it can result in long-term health risks such as heart disease, stroke, poor vision, poor circulation and kidney failure.
On the other end of the scale is hypoglycemia, or low glucose levels. When blood glucose levels fall below 75mg/dl, there is an immediate risk of losing consciousness, and even the risk of death. This occurs between meals as the body uses up glucose. During long periods of fasting, like at night, low glucose levels can be a big problem for diabetics.
Treating Diabetes
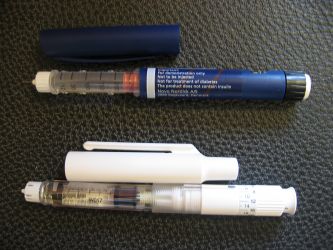
There are several ways to treat diabetes depending on the severity of the disease. Traditionally, doctors help their patients regulate glucose levels using exercise programs, a strict low-sugar diet, and insulin injections (see Fig. 2). It is not uncommon for a diabetic to take an insulin injection before a meal to help their body absorb glucose, thus avoiding hyperglycemia. Occasionally there is a problem of injecting too much insulin. In this case glucose is used up far too quickly and the scale tips in the other direction as glucose levels plummet and hypoglycemia sets in.
Knowing glucose levels is key when prescribing a treatment for diabetes. Traditionally, glucose readings have been taken using a finger stick method. Diabetics place a drop of blood from the fingertip into a well in the glucose sensor and it analyzes their glucose level at that moment. In addition to minor pain, this procedure is inconvenient and the patients rarely do it more than four times per day. When prescribing a treatment pattern for their patients, physicians look for trends in glucose levels in order to more accurately prescribe the correct treatment schedule. Four readings per day provide limited information. Continuous monitoring would show excursions from normal glucose levels.
Maintaining proper glucose levels is like walking a tight rope. While most of us have complex hormone systems that carry out this balancing act, people with diabetes have to pay close attention to their diet, exercise, and lifestyle. Even with very close attention to these things, diabetics can rarely keep their glucose levels stable enough to avoid long term health problems.
Glucose Sensor Chemistry
How is the glucose concentration of the blood actually found? There are many methods, including at-home finger sticks and intravenous catheters used at hospitals. Almost all of today’s glucose sensors incorporate some form of the Clark electrode and the enzyme glucose oxidase [3]. Most fundamentally, the Clark electrode detects hydrogen peroxide H2O2. It consists of platinum metal coated with glucose oxidase, an enzyme extracted from cows, and a few biological membranes. As blood diffuses across the biological membranes any glucose present in the blood reacts with glucose oxidase and is broken down into gluconic acid and hydrogen peroxide. The hydrogen peroxide then reacts with the platinum electrode and releases one electron for every molecule of glucose. Basically, the enzyme breaks down glucose forming hydrogen peroxide as a by-product. As the peroxide reacts with the metal it produces a current that is proportional to the concentration of glucose in the blood. The current is then converted into a glucose concentration.
Continuous Glucose Sensing
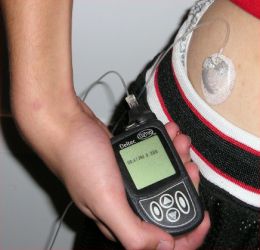
One of the most reliable methods of continuous sensing is the Continuous Glucose Monitoring System (CGMS) offered by MiniMeda. Unlike conventional sensors that take isolated glucose readings from the blood, the CGMS continuously detects glucose levels in interstitial fluid (the fluid which surrounds our cells).
This system uses a needle to insert a very narrow Clark electrode beneath the skin (usually on the abdomen) where it encounters interstitial fluid (see Fig. 3). This electrode is worn for 72 hours before it is replaced with a new electrode. Readings are acquired every 10 seconds and an average value is saved in a pager-sized monitor every 5 minutes. Every two weeks the data is downloaded onto a physician’s computer where the continuous glucose data can be viewed in graphical form. Their goal is to “help identify periods of significant glycemic excursion which would allow the physician to suggest specific changes in the timing and dosage of insulin infusion or injection” [2].
Knowing continuous glucose levels lets doctors treat diabetic patients better and allows the patients to follow a treatment program that is more personally tailored. In one case, doctors adjusted the therapy schedules of ten patients based on the information recorded by the CGMS. In only five weeks all patients showed a dramatic improvement in the consistency of their glucose levels and had very few excursions from the desired 75-120 mg/dl glucose range [1].
The advantages of the CGMS are many, including its reliability, safety, and availability. This device has full FDA approval and is available on the US market as the first “at-home continuous glucose sensor.” After calibration, this device is very accurate in its glucose detection, due its average number of 8640 readings every day. Additionally, patients experience very little irritation associated with this sensor. According to an in-house test, only 3 of 102 wearers complained of discomfort [1].
There are a few disadvantages to this system. Glucose values are not displayed to the wearer in real-time and there are no alarms to notify the wearer if their glucose level strays from the safe range. Instead the data is accumulated in the “pager” for two weeks before doctors download graphical data. Thus, the CGMS cannot be used for the immediate regulation of diabetes, only in overall program formulation. Another complication with subcutaneous sensors is due to the body’s own defense system. In the case of the MiniMed sensor, a needle prick is used to insert the sensor. As this wound heals, proteins and cells begin to coat the sensor and block the glucose from reaching the sensor; thus the sensor is good for only 72 hours before it must be replaced.
The Future of Continuous Glucose Sensing
Currently, doctors use continuous glucose data to formulate more personalized diabetic treatment programs, but the future will bring many other applications. For example, future versions of the CGMS will incorporate alarms that sound when glucose levels stray from the target range. Diabetics will be able to react immediately, thus avoiding the serious consequences associated with hyper and hypoglycemia.
Even more promising is the hope for artificial pancreas. Insulin pumps are widely used to deliver small amounts of insulin over a long period of time as opposed to large doses injected two or three times a day. This more closely simulates the body’s own insulin production and is very helpful in maintaining stable glucose levels throughout the day. Engineers at several institutions are working to pair insulin pumps with continuous glucose sensors to create, in essence, an artificial pancreas [4]. The glucose sensor will detect the level of glucose in the blood and then it will “tell” the pump to deliver the appropriate amount of insulin required to maintain a constant glucose level between 75-200mg/dl. Though this feedback technology has a long way to go, it offers much hope to diabetic patients. Currently doctors treat diabetes, but an artificial pancreas would be a significant step in the direction of curing the disease.
Conclusions
The field of continuous glucose sensing is rapidly growing. At this point, the MiniMed continuous glucose monitoring system is the only continuous glucose-monitoring device that has full FDA approval for use in the United States (though the Cygnus GlucoWatch ® has achieved partial approval). As technology in this field advances, continuous glucose monitoring will revolutionize the way we treat diabetes.
References
[1] J. Mastrototaro. “The MiniMed Continuous Glucose Monitoring System (CGMS).” Journal of Pediatric Endocrinology & Metabolism. vol. 12, pp. 751-758, 1999.
[2] B. Bode, T, Gross, K. Thornton, and J. Mastrototaro. “Continuous glucose monitoring used to adjust diabetes therapy improves glycosylated hemoglobin: a pilot study.” Diabetes Research and Clinical Practice. vol 46, pp 183-190, Sept 1999.
[3] G. Kost, H.T Vu, and J.H. Lee. “Multicenter study of oxygen-insensitive handheld glucose point-of-care testing in critical care/hospital/ambulatory patients in the United States and Canada.” Critical Care Medicine. vol. 26, pp 581-590, 1998.
[4] M. Gerritsen. “Problems Associated With Subcutaneously Implanted Glucose Sensors.” Diabetes Care by the American Diabetes Association. vol. 23, pp. 143-145, Feb. 2000.
[5] G. Pevere. “Infrared Nation.” The International Journal of Infrared Design, vol. 33, pp. 56-99, Jan. 1979.
[6] Medtronic. Medtronic MiniMed, Inc., Mar. 26, 2011.