At the time of writing, Meena Singh was a junior pre-medical student majoring in Biomedical (Biochemical) Engineering. She plans to earn her B.S. degree in May 2003.
Many women who are unsatisfied with the appearance of their breasts use augmentation mammioplasty, or breast augmentation, to achieve their desired look. The most commonly used breast implants are silicone gel and saline-filled implants, both of which have harmful drawbacks. Silicone gel are not biocompatible and obstruct the visibility of tumors in mammography. Saline-filled implants are found to harbor microbes that can cause infection. Because of these disadvantages, scientists have investigated the use of safer, natural alternatives to implant filler materials. The Trilucent soybean oil-filled breast implant was designed for increased safety, longer life, and radiolucency (ability to visualize tumors on a mammogram). These implants were thought to have additional advantages, such as increased biocompatibility and prevention of microbe growth. Additionally, the implant has a valve that allows surgeons to adjust its size after implantation and a microchip that allows doctors to track implant information. The Trilucent breast implant was supposedly the best implant design thus far; however, unpredicted complications arose in patients, causing the Trilucent implant to be removed from the market.
The Need for Breast Augmentation
Breast augmentation is the second most commonly performed cosmetic surgery in the United States [1]. Women who undergo this procedure typically want to enhance their body contour to increase their bust size, balance breasts that are different in size, or correct a reduction in breast volume after pregnancy. Over the past 30 years, nearly two million women have received breast implants in North America [2]. In recent years, breast implants have been in particularly high demand [1].
Background of Breast Implants
The optimal breast implant should be nontoxic, noncarcinogenic (non cancer-causing), sterilizable, biocompatible, radiolucent (able to visualize tumors in a mammogram) and relatively affordable [3]). Since the introduction of breast prosthesis in 1962, silicone gel-filled implants (see Fig. 1) have been the dominant implants; yet they are neither biocompatible nor radiolucent [3]. Saline-filled implants are also radiopaque on mammograms, making it difficult to recognize tumors [3]. Additionally, breast implants can eventually rupture, releasing a few cupfuls of fluid into the breast tissue. In saline-filled implants, this fluid might harbor a wide variety of potentially harmful microbes. These microbes can infect the tissue surrounding these implants, squeezing the area around the implant, and causing the breasts to harden and contract [4].
Soybean Oil-Filled Breast Implants
Because of the unsafe drawbacks of silicone gel and saline-filled implants, scientists have investigated safer, natural alternatives for filler materials. In the 1980’s, scientists at Washington University developed the Trilucent soybean oil-filled breast implant (Lipomatrix, Neuchatel, Switzerland). It was implanted in about 10,000 European women in 1994 and 1995 [5]. The soybean oil-filled breast implant functions like a saline-inflatable implant, but was engineered for radiolucency, safety, and longer life.
Properties of the Filler Material
The Trilucent breast implant is filled with Trilipid Z6, which contains highly refined medical-grade triglyceride fats extracted from soybean oil [6]. A triglyceride is a macromolecule (complex molecule composed of smaller molecules) containing three long fatty acid chains bound to a molecule of glycerol. The triglycerides in Trilipid Z6 are composed of 60% polyunsaturated fatty acids, 26% monounsaturated fatty acids, and 14% saturated fatty acids [5].
Biocompatibility
Biocompatibility is achieved when the body trusts a synthetic material as a normal constituent and the material undergoes normal metabolic degradation. Because breast tissue is composed of fatty tissue, Trilipid Z6 was designed to be nearly identical to body constituent and normal dietary fats to enhance the biocompatibility of the filler material. In case the shell leaks or ruptures, it is believed that the filler material will be absorbed and metabolized by the body and either redistributed to the natural fat stores in the body or excreted in the urine [7].
Triglycerides from soybean oil have shown biocompatibility in both humans and animals. The filler did not show a toxic or allergic response when injected in rabbits; it did not initiate an immune-mediated response associated with foreign materials in the body [7]. Additionally, triglycerides of medical-grade soybean oil have been used medically for over 40 years as intramuscular drug carriers and as intravenous nutrition for critical care patients [5].
Growth of Microbes
Another advantage of the soybean oil-filled breast implant is that unlike saline-filled implants, triglycerides do not support fungal or bacterial growth, and bacteria rarely inhabit the tissue surrounding these implants [5]. This is because small leaks from the implant kill neighboring microbes. Since soybean oil-filled implants do not harbor microbes, capsular contracture, in which growth of microbes around the implant squeeze and harden the implants, is prevented.
Radiolucency
Breast cancer develops in about 10% of the women with breast implants, so it is very important to be able to detect masses on a mammogram [3]. Thus, one of the major advantages of using triglycerides as a filler is the radiolucency of the material. Radiolucency is determined by a material’s effective atomic number, which is the relative amount of photoelectric interaction between different materials and a radiation beam; the resulting amount of interaction produces the contrast in radiographic images [7]. Because saline and silicone implant fillers have atomic numbers that are higher than that of the breast tissue, they are radiopaque on a mammogram [7]. Soybean oil filled implants, however, are radiolucent on a mammogram because they have an effective atomic number near that of breast tissue [7].
Viscoelasticity
The viscoelasticity of Trilipid Z6 causes the material to feel similar to natural breast tissue, permits the breast implant to move naturally, and causes it to remain soft without capsular contracture. It also prevents the wrinkling that may occur in saline-filled implants [7]. Additionally, triglycerides, unlike saline, are an excellent lubricant. This decreases the amount of abrasion with the shell, reducing leakage and rupture [7]. Therefore, the Trilucent breast implant is expected to have a longer life than saline-filled implants.
Properties of the Shell
The Trilucent breast implant has a heat-cured silicone elastomer shell. Aging of the shell occurs as soon as the prosthesis is implanted, so it is important to understand its composition and durability.
Composition
The uncured silicone elastomer shell is produced by mixing polydimethylsiloxane with a fumed amorphous (non-crystalline) silica filler and a polymerizing agent [8]. The fumed silica is used to enhance the mechanical properties and increase tear resistance.
Mechanical Properties
The Trilucent implant was engineered for longer life, a property directly influenced by the mechanical properties of the shell, such as tensile strength, tear resistance, and shell thickness. The minimal tensile strength required of the shell by the Food and Drug Administration is 900 pounds per square inch, and the tensile strength of the Trilucent breast implant shell exceeds this with 1038 pounds per square inch [8]. The shell was also found to have a minimum tear resistance of 99 pounds per inch, which is also above the minimal requirement. Additionally, the shell is .0165 inches thick, which is optimal for both elasticity and strength. Therefore, the shell is designed to reduce the risk of implant rupture and leakage due to improved abrasion and tear resistance.
Surface of the Shell
Micro structured pillars are integral parts of the implant shell because they create a textured or roughened surface. Texturized implants are believed to reduce capsule contracture, preventing scar tissue from growing around it smoothly [9].
Valve
A unique feature of the Trilucent implant design is a small valve on the implant surface. The valve facilitates insertion of the implant into the body because it allows surgeons to inflate the implant after it is placed in the body; this decreases the risk of damage to the surrounding breast tissue and pectoral muscle. Additionally, the valve allows adjustment of the size after implantation by increasing the amount of oil in the implant.
Microchip
Another innovative aspect of the implant is the inclusion of a tiny microchip on the center surface of the implant. The microchip provides doctors with noninvasive identification of the implant in case there is a problem in the future; it is also capable of storing the patient’s history.
Electronic Components
The electronic components of the transponder are encapsulated in a sealed glass tube made of biocompatible soda lime glass. The three major components inside the tube are the silicon computer chip, the copper wire, and a capacitor.
Silicon Computer Chip
The silicon computer chip is a custom integrated circuit that contains the chip’s unique ID code.
Copper Wire
The coil of copper wire is wound around an iron core rod. The coil acts as both an antenna and as a transformer induction system that converts the radio-frequency wave from the scanner into electricity to power the chip. Therefore, since the chip is passively powered without a battery; it can never wear out.
Capacitor
The capacitor stores energy and assists signal transmission to and from the microchip.
How the Transponder Works
The chip and radio-wave scanner uses low frequency radio signals that can penetrate the breast tissue. Once the scanner is activated, it displays the chip’s ID number on a liquid crystal display screen. Then the scanner transfers the chip ID codes to a computer database which accesses the implant and patient information.
Augmentation Mammioplasty
Incision Sites
The first step in this procedure is to decide on the implant size. Then, the incision site is determined; this can take place around the darker part of the nipple (areolar), under the armpit (transaxillary), under the breast (inframammary), or through the navel (transumbilical) [10]. Each site has its advantages and disadvantages. For example, in the areolar incision, the scar is masked by the pigmented part of the nipple. The inframammary site, however, leaves a more visible scar. The transaxillary is the most hidden of all sites because scarring only occurs under the armpit, and the transumbilical incision site may only leave a scar by the navel.
Implant Placement
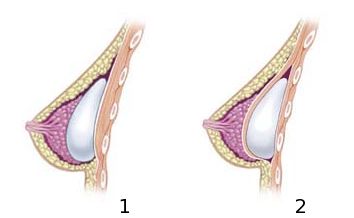
The implant can be placed either above (subglandular) or under the muscle (submuscular) [10]. In the subglandular position, there is less post-operative discomfort because only skin and fat are cut. However, in this position, there is high obstruction on a mammogram, rippling of the skin over the implants, and a higher risk of capsular contracture [11]. Nevertheless, the only way to use the submuscular position without cutting the muscle is through the transaxillary incision because the implant can enter under the muscle. Some advantages of this approach are low interference on a mammogram, no visibility of the implant, no rippling effect, lower risk of capsular contracture, and no scars on the breast [11]. However, there is more post-operative muscular discomfort [11]. In any method, the implant is placed in the desired position, and the surgeon uses the implant valve to increase the amount of oil in the implant and obtain the desired size and look (see Fig. 2).
Potential comlication with translucent implants
In any augmentation mammioplasty procedure, there are a number of potential complications. Trilucent implants were removed from the market in 2000 because in some women they were found to cause red and painful breasts from serious infections [12]. This was due to leaking oil that emulsified in the breast and turned rancid. Additionally, Trilipid Z6 was found to break down into aldehydes, genotoxic chemicals that can damage DNA in the cells and cause cancer. [12]. Women with Trilucent implants were advised to avoid pregnancy until the implant was removed because leakage or rupture of the implant would provide increased exposure to the aldehydes [12].
Conclusion
The Trilucent soybean oil-filled breast implant was supposed to be the innovative natural filler material that overcame the disadvantages of the more commonly used silicone gel and saline-filled implants. They were engineered and designed to be a biodegradable and biocompatible material, a lubricant, a more viscous fluid than saline, a nonallergenic and radiolucent filler, and a normal dietary component. However, this new and improved Trilucent implant turned out to have some serious complications. These unpredicted problems were so severe that the implant was removed from the European market. Thus, it seems that scientists have yet to design a completely safe breast implant to allow women to achieve their desired physical appearance.
References
-
- [1] L. Bren. “Saline Breast Implants Stay On Market As Experts Warn About Risks.” FDA Consumer, pp. 34, Jul. 2000.
- [2] W. Immen. “3 Canadians get breast implants filled with special soybean oil.” Toronto Globe and Mail. Feb. 1996.
- [3] D. Monstrey et al. “Biocompatibility and Oxidative Stability of Radiolucent Breast Implants.” Plastic & Reconstructive Surgery, vol. 105, pp. 1429, 2000.
- [4] C. Woolston. “Saline-filled breast implants can harbor microbes.” Washington University Record. Sept. 1997.
- [5] K. Weingartner. “What is Trilucent Breast Implant?” University of Illinois Soybean Utilization Williams, Gary. “Assessment of Health Risks Associated with Exposure to Soybean Oil Filler Compounds and Peroxidation Products Potentially Released from Trilucent Breast Implants.” Trilucent Info Website. Oct. 22, 2001.
- [6] M. Barnett. “Triglyceride-filled Breast Implants.” Plastic & Reconstructive Surgery, vol. 99, pp. 2105-2106, 1997.
- [7] W. Brucker and L. Sendelbach. “Bleed of and Biologic Response to Triglyceride Filler Used in Radiolucent Breast Implants.” Plastic & Reconstructive Surgery, vol. 99, pp. 2108, 1997.
- [8] Y. Brandon, et al. “Variability in the Properties of Silicone Gel Breast Implants.” Plastic & Reconstructive Surgery, vol. 108, pp. 647-655, 2001.
- [9] B. Bern and J.W. May. “Biophysical and Histologic Properties of Capsules Formed by Smooth and Textured Silicone Implants.” Plastic and Reconstructive Surgery, vol. 89, pp. 1037-1042, 1992.
- [10] A. Richards. “Breast Augmentation.” The Birth to Venus Website 21 Oct. 2001. “Testing to Begin on New Breast Implant.” Medical Materials Update. 1, pp. 47, 1994.
- [11] T. Dewire. “Anatomy of Chest Wall and Breast Implant Placement Over or Under the Muscle.” Advanced Art of Cosmetic Surgery Website. [On-line]. Available: http://www.advanced-art.com/Breast-Augmentation-Info.htm [Oct. 23, 2001].
- [12] D. Zuckerman. “Are Breast Implants Safe?: The Importance of Empirical Research.” Medscape. [On-line]. Available: http://www.medscape.com/viewarticle/408187_5.
- [13] E. Ramesh. “Time Enough? Consequences of Human Microchip Implantation.” Franklin Pierce Law Center Website. [On-line]. 22. Available: http://www.fplc.edu/risk/vol8/fall/ramesh.htm [Oct. 2001].