Key Words: Quantum Dot Display | Quantum Dot Light Emitting Diode | Virtual Reality | Semiconductors |
Abstract:
Quantum mechanics is often perceived as a theoretical science that is outside the scope of our everyday lives. However, it is the driving factor behind the creation of semiconductor materials that allow for increasingly accurate virtual and augmented reality displays. Quantum Dot Light Emitting Diodes (QLEDs) provide the tools for enhancing the vivid realism of video game consoles and scientific simulations. Investigating the physics principles behind QLEDs allows for an understanding of the technology that is paving the path into our virtual future.
Your Virtual Reality Vacation:
It has been a long day at work, and you are enjoying some well-deserved relaxation. You gaze at the breathtaking view in front of you: calm, teal blue waves gently lapping a soft, caramel-colored beach. The sun is just disappearing over the horizon, illuminating the whole scene in a golden-red glow, and accentuating the shadow cast by each individual grain of sand. Despite the dusk, every detail is in sharp focus. From the seagull waddling after a clumsy crab to the sand dollar that just washed up on the shore, the scene is one of unforgettable vivacity.
Despite your bliss, you know you must make dinner and take the dog for a walk. You slip the virtual reality glasses off your face and the ocean sunset is replaced by your bedroom window and dresser. The picturesque trip to the beach was just what you needed to get back on your feet.
Virtual reality (VR) and augmented reality (AR) are becoming increasingly prolific and accurate in their depiction of 3D visuals. VR headsets are common in video games, theme park attractions, and architectural design evaluations. With enhancements in optics and display technology, VR is on track to provide exceptionally realistic virtual vacations. But what is the technology behind these crisp images?
Quantum Dot Light Emitting Diodes (QLEDs) are a new semiconductor technology that use quantum mechanical principles to precisely control the color and amount of light visible on a display.
Currently, organic LEDs (OLEDs), which are LEDs made from organic compounds, are the primary method used for lighting the screens in smartphones, TVs, tablets, laptops, and monitors. These optoelectrical devices utilize the unique chemical properties of semiconductor materials to allow electrons to transition from higher to lower energy states when connected to a battery or electrical outlet [1]. When an electron’s energy is lowered, energy is released from the semiconductor in the form of light, which is what you observe coming from your screen.
Unlike OLEDs, new QLEDs can be precisely constructed to control the color of light emitted from a screen, thereby providing a much wider spectrum of colors that more closely approximates those seen by the human eye [2].
Quantum mechanics is the secret behind the exceptional photorealism we expect to see with QLED VR/AR displays. The light produced by a QLED is defined by the energy of electrons within a semiconductor material. It is possible to force an electron to have a specific energy by placing it into a “trap” that confines the electron to that energy. The energy of the electron is determined by the size of the trap. When the electron’s energy changes from high to low inside of the trap, energy is emitted as light. The color of light emitted is determined by its energy. Therefore, setting the size of the trap allows you to precisely determine the color and type of light emitted from a QLED.
This fine-tuned control allows for the sophistication of QLED technology. To gain an understanding of how quantum dots enhance the possibility of virtual reality landscapes, we will explore the concepts of quantum mechanics using the analogy of being on a virtual vacation.
Your Vacation Itinerary
DAY ONE
8:00 AM: Bungee Jumping in LA (Understanding Energy)
The first step is to understand the energy of an electron. On the first morning of your virtual vacation, you go bungee jumping from the top of a skyscraper in downtown Los Angeles. As you stand on top of the building, you have some stored energy (potential energy) due to gravity because of your height above the ground (Fig. 1). When you jump off the building, your stored energy becomes motion energy (kinetic energy) (Fig. 2).
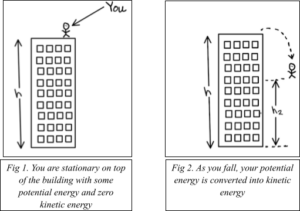
An electron in a trap also has some potential and kinetic energy. The potential energy of the electron is analogous to the gravitational potential energy you experience while bungee jumping from a building. But how do you trap an electron to give it some energy in the first place?
2:00 PM: Mini-Golfing (Creating a Trap)
An electron can be trapped by placing it into a “hole” that it cannot get out of. This leads you to the next step in your vacation. After virtual bungee jumping, you decide you need something less terrifying to occupy your afternoon. You decide to play mini golf (the QLED display you are using is so good that this golfing experience is far better than playing golf on your average Wii). In this example, your golf ball falling into a hole is analogous to an electron becoming trapped in a semiconductor material. 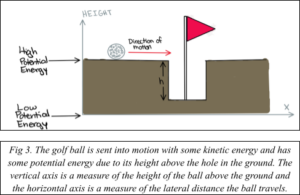
As you hit the ball toward the first hole with your golf club, you give it some initial kinetic energy (Fig. 3). You are a highly skilled mini golfer, so your ball falls into the hole on the first try (Fig. 4). The potential energy of the ball decreases and is converted to kinetic energy as it falls.
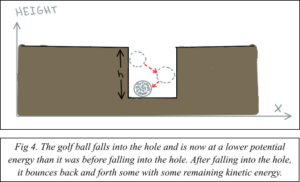
The kinetic energy that the ball has as it bounces around in the hole can be any number depending on how hard you hit it. Eventually, the ball will lose energy due to friction and become stationary. Now your ball has a new potential energy that is lower than it was at its start.
Before moving to the next hole, you need to retrieve your ball. It would not make sense for the ball to spontaneously come back up to the surface; it is at a lower energy state than it was initially. In other words, the hole is an energy barrier that the ball has become confined to. This is because the energy of the ball inside the hole is lower than it was outside of the hole.
The same principle applies to an electron that is trapped in a semiconductor. This trap is called a quantum dot.
6:00 PM: Dinner at Schrödinger’s Café (Setting a Size)
Now that you know how to trap an electron, your next objective is to give the electron a specific energy (ex: high, low, intermediate). To do this, you need an electron trap with a specified set of dimensions. It happens that the next activity on your itinerary is to stop for some virtual dinner at Schrödinger’s Café, the perfect place for ordering custom electron traps.
We are digressing slightly from the golf example. While the potential energy of an electron is like that of a golf ball, an electron trap is not a physical hole as is the case for the hole trapping the golf ball. Instead, the electron trap is created by the difference in energy of two different semiconductor materials. This is analogous to the height difference of the golf ball when it is above the ground and then inside of the hole.
Additionally, an actual electron cannot be represented as a single solid object like a golf ball. Instead, an electron behaves more like a wave (like a sound wave or a ripple in a pond). However, an electron can still be trapped in an energy barrier due to its existence in a lower energy state, just as your golf ball was trapped in the hole because it had a lower potential energy than where it started.
At Schrodinger’s Café, a waitress shows you to your seat and gives you a menu. The menu only has one dish:

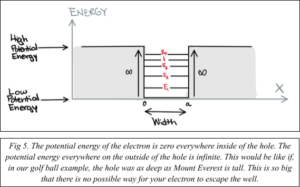
You tell the waitress that you would like to order the house combo with an electron trap (1D quantum dot) that comes in two sizes: one that is 200 nm wide and a second one that is 400 nm wide (Fig. 6). (Note: A nanometer is one billionth of a meter. This is about 200 times smaller than the width of a strand of hair.)
The energy of the electron is determined by the size of the electron trap. A wider 1D quantum dot means the electron has a lower energy, while a narrower 1D quantum dot means the electron has a higher energy.
Not only can you use one equation to solve for the state of your electrons at various energy levels, but you can also decide exactly what those energy levels are by making your 1D quantum dot a particular size! The final steps in your virtual vacation journey will lead you to understand how to use your 1D quantum dot to produce colored light.
DAY TWO
7:00 AM: Wake Up (Emitting Light)
As you wake up from a night of blissful sleep, you slip on your virtual reality headset to be greeted by a glorious morning sunrise over the ocean.
This reminds you that you still need to know how a 1D quantum dot emits light. Luckily for us, our electron can shift to and from higher or lower energy levels (Fig. 7).
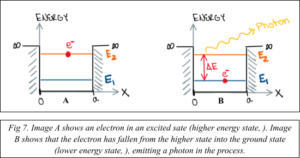
First, we consider that our electron is in a high energy state, called an excited state (Fig. 7 A). The electron may then “fall” into a lower energy state, called the ground state (Fig. 7 B). The law of conservation of energy states that energy must be conserved, and it cannot be created or destroyed. If the electron loses energy when it transitions to the ground state, where did that energy go? In this case, it was released in the form of a photon, which is a particle of light. The energy of this photon is exactly equal to the quantized energy difference between the electron’s excited state and its ground state.
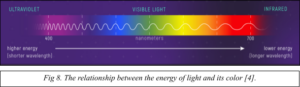
The energy of the released photon is directly related to the wavelength of the emitted light, which determines the color of the light (Fig. 8). Recall your order from Schrodinger’s Café. The small 1D quantum dot (200 nm) contained higher energy electrons, which would lead to the emission of higher energy photons. High energy visible light is blue and violet. The large 1D quantum dot (400 nm) contained lower energy electrons, which would lead to the emission of lower energy photons. Low energy visible light is red and orange.
This is the basic principle behind the emission of visible light from a QLED.
8:00 AM: Making A Peanut Butter Sandwich for Breakfast (Semiconductor Construction)
Now you want to make your own QLED. You decide to make yourself a virtual peanut butter sandwich for breakfast (no jelly because peanut butter is healthier). This sandwich represents your QLED. The bread on either side of your sandwich represents one type of semiconductor material. The peanut butter represents the second type of semiconductor material. Electrons in the peanut butter semiconductor exist at lower energy states than when they are in the bread semiconductor.
The semiconductors in your peanut butter sandwich only allow for an electrical current to flow in one direction [5]. A device that only allows electrical current to flow in one direction is called a diode. The semiconductor materials are created by taking a normally insulating substance (which prevents the movement of electrons) such as silicon and injecting it with impurities containing conductive substances (which allows the movement of electrons). By adjusting the ratio of conductive to insulating material, a semiconductor is designed to carefully control the motion of electrons.
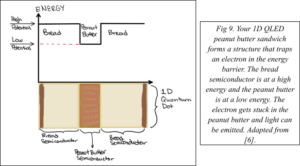
For a QLED, electrons pass through the sandwich and get trapped in the low-energy peanut butter semiconductor such that your sandwich has become a 1D quantum dot (Fig. 9). The peanut butter region of the LED is the optically active region, that is, photons of light are emitted directly from this part of the device and are seen as visible light [7]. The width of your peanut butter layer determines the energy and color of light emitted.
In an OLED or a non-quantum dot LED, there is no middle layer of peanut butter material that allows for an electron to be trapped. Instead, there might simply be an energy difference between two material types (emissive and conductive layers) that can emit light. It is far more difficult to control the color of light emitted from this type of LED than for a QLED.
END OF YOUR VIRTUAL VACATION ITINERARY
Conclusion: The Advantages of QLEDs
The ability to trap an electron at precise energies to customize the color of a quantum dot provides many advantages in display applications [8]. QLED displays provide a much broader spectral window (range of possible colors) and are more chemically versatile compared with standard commercialized high-definition television displays [8]. The ability to precisely control the color of light allows the display to maintain wide arrays of colors at high accuracy while at peak brightness, avoiding the potential for washed out colors, grainy textures, or burn in that can accompany OLED displays [2][9]. While a typical OLED display may combine three standard colors (red, green, and blue) to produce all other colors visible on a screen, a QLED display allows for nearly every pixel to produce the precise color required at that point, whether it be indigo, violet, chartreuse, or scarlet.
Additionally, engineers have optimized the design of QLEDs to be as small as possible to avoid reabsorption of emitted photons while still confining an electron [7]. This means that QLEDs can be packed extremely close together on a display, providing an exceptionally high amount of light per surface area of a screen [9]. This optimization also maximizes the efficiency of QLEDs as light sources, making them potentially more energy efficient than traditional OLEDs [7].
This could help to satisfy the demand for maximum realism and higher resolution displays with the expansion of AR and VR applications [8]. The uniquely customizable optical properties of QLEDs will allow for the perfection of features such as better angular resolution and a wider field of vision (FOV) while aiding in the mitigation of issues related to accommodation-vergence conflicts and hard-edge occlusion [10]. This means producing displays with nearly perfect 360-degree visual accuracy while allowing the viewer to focus on 3D objects with varying levels of depth perception in a way that makes them appear hyper-realistic [10].
Overall, QLEDs are forging a new path towards ultimate realism in AR and VR, making it ever more possible for you to explore global tourist attractions or ride a dragon through a fantasy kingdom all from the comfort of your couch. So, when the first quantum dot televisions and VR headsets hit the shelves, be sure to check them out.
Links and Books for Further Reading:
- Understanding the difference between virtual and augmented reality: What’s the Difference Between AR and VR? | Tulane School of Professional Advancement
- Understanding the Future of VR and AR in terms of Entertainment and Vacation Destinations: How Virtual Reality (VR) is Transforming the Travel Industry | Revfine.com
- Novel by a bestselling author on understanding quantum mechanics: Quantum Mechanics, The Theoretical Minimum: What You Need to Know to Start Doing Physics by Leonard Susskind and Art Friedman
Additional Multimedia Resources for Further Exploration:
- How LED works | What is a LED (Light Emitting Diode): How LED works ⚡ What is a LED (Light Emitting Diode) – YouTube
- Jordan Edmunds is a fourth-year Ph.D. candidate in EECS department at UC Berkley working on nanofabrication, electromagnetics, and electronics. He has a YouTube series of highly informative videos on electromagnetism, quantum wells, and diodes: Jordan Edmunds – YouTube
- What are Quantum Dots?: What are Quantum Dots? – YouTube
References
[1] CircuitBread, J.B. Magoncia, United States. How Does an LED Work at the Molecular And Practical Level? | Intermediate Electronics. (Sep. 19, 2019). Accessed: Aug. 30, 2022. [Online Video]. Available: https://www.youtube.com/watch?v=No8PZsLnjZU
[2] D. Haynes. “What is Quantum Dot display technology?” Insights. https://insights.samsung.com/2021/12/29/what-is-quantum-dot-display-technology/ (accessed Sep. 18, 2022).
[3] D. Griffiths and D. Schroetter, “Time-Independent Schrӧdinger Equation,” in Introduction to Quantum Mechanics, 3rd ed. Cambridge, United Kingdom: Cambridge University Press, 2018, ch. 2.
[4] L. Hustak. “Relationship Between Color, Wavelength, and Energy.” Webb Space Telescope. https://webbtelescope.org/contents/media/images/01F8GF6NH5PTXGBVNVHR2TSQRB#:~:text=Different%20colors%20of%20light%20have,violet%20end%20of%20the%20spectrum (accessed Dec. 11, 2022).
[5] VirtualBrain [ENG], E. Piñones. How LED works | What is a LED (Light Emitting Diode). (Jun. 30, 2021). Accessed: Aug. 31, 2022. [Online Video]. Available: https://www.youtube.com/watch?v=9BDTtcRMxpA&t=237s
[6] CMS College Kottayam, Jeya P. Photonics-1, Mod1, 1.6Absorption in Quantum wells |Jeya P| Department of Physics. (Jun. 30, 2020). Accessed: Aug. 21, 2022. [Online Video]. Available: https://www.youtube.com/watch?v=p1iOt6MCjFA&t=416s
[7] S. Cronin. University of Southern California, Ming Hsieh Department of Electrical and Computer Engineering. (Aug. 3, 2022) Interview on Quantum Mechanics and LEDs. [Live Interview Source].
[8] M.K. Choi, J. Yang, T. Hyeon, and D.H. Kim Eds., “Flexible quantum dot light-emitting diodes for next-generation displays,” in npj Flexible Electronics 2, Apr. 5, 2018. [Online]. Available: https://doi.org/10.1038/s41528-018-0023-3
[9] “Quantum Dot Ink for Augmented & Virtual Reality, Displays”. Chain Reaction Innovations Entrepreneurship at Argonne. https://chainreaction.anl.gov/quantum-dot-ink-for-augmented-virtual-reality-displays/ (accessed Sep. 18, 2022).
[10] H.J. Jang, J.Y. Lee, J. Kim, J. Kwak and J.H. Park, Eds., “Progress of display performances: AR, VR, QLED, and OLED, Volume 21 – Issue 1,” in Journal of Information Display, Feb. 2020. [Online]. Available: https://www.tandfonline.com/doi/full/10.1080/15980316.2020.1720835



