Sunglasses are often taken for granted as just another fashion accessory, but they are actually the result of engineering technology that is both powerful and delicate. In order to create a final product that is protective, stylish, and durable, engineers were forced to deal with the complex nature of light — especially ultraviolet radiation and polarization — and the way people view the world. This fine line between form and function has resulted in a final creation that is appealing from both a visual and scientific standpoint — it protects your eyes and looks “cool” at the same time.
Introduction
Jacqueline Kennedy Onassis. Tom Cruise in “Risky Business.” The Blues Brothers. The cast of “Reservoir Dogs.” In modern popular culture, the images of these stars are linked by one of the most important safety devices ever developed. It just happens to be a stylish fashion accessory as well.
Sunglasses seem like simple inventions – just two pieces of tinted glass that cover your eyes. But modern sunglasses have two distinct and important requirements: to protect your eyes from the harmful elements of sunlight and to look fashionable. Herein lies the engineering challenge: to design technologies that can conquer the complex and potentially dangerous nature of light while remaining aesthetically pleasing. In order to fulfill these complex objectives, engineers have developed lenses and filters that counter the harmful effects of UV rays, as well as high-tech, bendable metal frames to safely house them (see Fig. 1). Modern sunglasses are true combinations of form and function.
Beyond Style: Why We Need Sunglasses
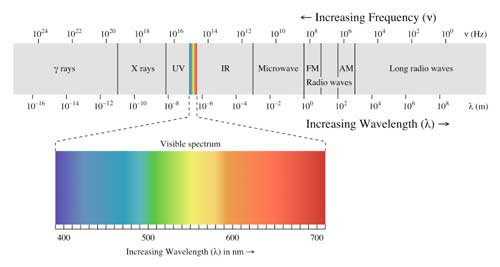
The complexity of sunglass technology is dictated by the nature of sunlight itself. When you think about sunlight, you are most likely imagining the range of colors that leave the sun and end up reaching your eye. It turns out that the sun emits more types of waves and energy than those that you can see. Visible light is just a small portion of what is known as the electromagnetic spectrum.
The electromagnetic spectrum is a system that classifies the energy of electromagnetic waves by their wavelengths (See Fig. 2). The spectrum, ordered from longest to shortest waves, is composed of radio waves, microwaves, infrared waves, visible light, ultraviolet rays, X-rays, and finally Gamma rays. The sunlight that reaches the earth consists of waves with wavelengths between 100 and 1,000,000 nanometers – ultraviolet, visible, and infrared waves [1]. The infrared waves are responsible for the heat we feel from sunlight, and the visible waves are responsible for what we see. It is the ultraviolet radiation waves that we – and our eyes – have to worry about.
UV Rays and How They Are Stopped
When exposed to excessive levels of ultraviolet radiation, the eye – specifically the cornea and retina – can be severely damaged. If the exposure is intense enough, cataracts and macular degeneration may result, which can eventually lead to blindness [1]. For this reason, all modern functional sunglasses have UV protection.
The process by which waves are “stopped” is called absorption. Consider an example involving the visible spectrum: the contrasting experiences of wearing black versus white shirts. Certain materials, like the colored cotton of the shirts, absorb specific colors based on wavelength. The black shirt has properties that cause it to absorb the entire visible spectrum, resulting in a greater concentration of energy and an increase in heat, while a white shirt absorbs none of the visible spectrum and remains cool.
In much the same way, sunglass lenses are treated with an extremely thin chemical coating whose sole purpose is to absorb rays with wavelengths between 100 and 400 nm – the range for ultraviolet rays. While the exact chemical formula for this coating is a heavily guarded trade secret [2], the American Optometric Association requires that these ultraviolet absorbers block UVA and UVB radiation almost entirely – 1% transmission or less is the requirement to receive their Seal of Acceptance, a necessary certification in the modern retail world of sunglasses [3]. This certification is your guarantee for eye protection; dark-colored lenses alone do not guarantee safety. In sunglasses lacking proper UV protection, darker lenses cause your pupils to dilate, allowing for even more harmful rays to enter your eye than usual – this does more damage than if you were not wearing sunglasses at all.
Tints: More Than Just Colors
Once base UV protection is accounted for, people move on to what they generally consider a simple stylistic choice – the tint of the lenses. However, the tint you choose for your lenses should not just be a matter of personal preference, because different colors not only affect the way your eyes interpret the visual world, but also provide different levels of protection.
Since blue and violet have the shortest wavelengths in the range of visible light, and are therefore closest to ultraviolet waves on the electromagnetic spectrum, these colors can also result in damage to your eyes [1]. One way to counteract this effect is to wear yellow or amber sunglasses. These are often called “blue-blocking” lenses because, much in the same way that the UV coating works, they absorb potentially harmful blue visible light and thus provide extra protection. Unfortunately, since blue has such a short wavelength, it bounces around quite easily and has a great effect on many parts of the visual world [4]. Eliminating blue light therefore causes a great deal of color distortion, resulting in a trade-off: the so-called “blue blockers” are a good choice for increased UV protection, but a poor choice for accurate color recognition.
Many other tints also change perception, but without the added benefit of extra protection from short-wavelength rays. Green tints filter out much of the blue, and since the blue bounces everywhere, its elimination increases contrast, making green a good choice for sporting activities. For a more standard experience, grey tints provide good protection with minimal color distortion, making them a good choice for everyday activities [5].
The End of Glare: Polarization
Sunglasses must also prevent the problem of glare. This annoyance, which can become dangerous while driving, is a result of a phenomenon called polarization, which depends on light’s interaction with its surroundings.
Light waves can be thought of as plane waves, which are waves that move forward with velocity, but also propagate infinitely in all directions. When light is reflected off a surface – like the hood of a car or the surface of a lake – all the light that bounces off is forced to match the shape of the surface. This results in all the light waves being horizontal, increasing the intensity and causing glare. Such intense light is uncomfortable to the eye [6].
In 1926, after years of studying polarization, scientist Edwin H. Land developed the polarizing filter, a thin film that consists of many long, narrow, microscopic crystals aligned in a parallel manner [7]. When light is shined through one of these filters, only the waves that are perpendicular to the direction of the crystals pass through. All parallel light is absorbed. Thus, when light that is already horizontally polarized comes into contact with a horizontally polarized film, none of the light passes. This filter eliminates the issue of glare, and is still used in modern sunglasses over 80 years after its invention (See Fig. 3).
Bend But Do Not Break: Frames
Once the layers of protection on the lenses have been set, one final consideration remains: the frame. The experience of having your sunglasses sat on or dropped so hard that the frame is twisted or bent beyond repair is far too common. Luckily, scientists and engineers have employed a new type of metal in eyeglass frames – a “memory metal” – that allows these particular frames to bend and twist into nearly unrecognizable forms and then perfectly snap back to the original shape.
The key to this technology is the metal itself, an alloy called nitinol that consists of 55 percent nickel and 45 percent titanium [8]. This type of metal has two phases: the austentitic (high temperature) phase and the martensitic (low temperature) phase. The metal can be bent and twisted quite seriously in the martensitic phase, but once it is heated up to the austentitic phase, it moves back to its original shape [9].
The austentitic phase is developed when the metal is heated to approximately 500 degrees Celsius. At this temperature, the very atoms of the metal rearrange themselves into a cubic structure of extremely high symmetry. At room temperature, the metal is back in its martensitic phase, but for nitinol, a very small difference in temperature separates the austentitic and martensitic phases. Thus, the act of simply bending the frames is enough to move the temperature into the austentitic phase, thereby causing the frame to return to its proper shape almost instantly. Nitinol is typically used in the bridge, top bar, and temples of the frame, since this is where flexibility is needed most [9]. The combination of strength, flexibility and strategic placement results in nearly indestructible sunglasses.
A Future So Bright
Although the bar for modern sunglasses has been set very high, there is always room for future developments. As the general public ages and realizes the importance of eye safety, demand for a universal standard of 100% UV protection is just around the corner. Memory metals are getting better and better at “remembering.” But at this point, we see the start of a slight shift in sunglasses research and development, as scientists and engineers recognize that the modern pair of sunglasses is a remarkable achievement that is becoming more and more difficult to improve upon. They are instead rushing to integrate sunglasses into the more “traditional” engineering world, outfitting them with wireless connections to cellular phones and digital media players. As sunglasses become more high-tech and connected, developers should be sure to remember the fine balance of safety, function, and style that made sunglasses beautiful pieces of engineering in the first place.