Invented in 1983, the neodymium magnet, though not well-known, is the world’s most powerful permanent magnet. Composed of approximately 70% iron, 5% boron, and 25% neodymium, neodymium magnets are utilized in electric motors, smart phones, hard drives, headphones, speakers, and many more applications. Compared with its predecessor, the Samarium Cobalt magnet, it is 1.5 to 2 times stronger with less density. Recently, China, the biggest producer of neodymium, has limited exports, causing concerns of shortages. This has caused resurgence in domestic mining and raised possibilities of reduced dependence on foreign imports, hopefully attracting attention to limited resources.
Introduction
A modern-day CEO can be found cruising through town in his hybrid vehicle, typing away on his smart phone while listening to music through his headphones. All of the items he utilizes contain one common element: the neodymium magnet. The neodymium magnet was invented in 1983 [21] but has not received much attention. Even though these magnets are a crucial part of electronics today, many people have no idea what they are or what their purpose is. Invented and developed by General Motors and Sumitomo Special Metals, neodymium magnets, or NIB magnets, are made up of 70% iron, 5% boron, and 25% neodymium [2]. The key ingredient in this compound is the rare-earth metal neodymium, its namesake. Together, these three elements form the strongest type of permanent magnet known to man. The magnets are strong enough to hurtle through the air and break fingers. However, despite their inherent dangers, utilized properly, neodymium magnets can be extremely useful to society.
History
The neodymium (or neo) magnet was created because the previous industry standard of the Samarium Cobalt (SmCo) magnet was becoming too expensive. A more cost-efficient magnet was needed. Neo magnets were simultaneously discovered and created in 1983 by General Motors and Sumitomo Special Metals [1]. The two companies utilized different processes in creating the magnet. General Motors created them in a powder-bonded form while Sumitomo made them in a sintered form. These two different methods of production resulted in slightly varied properties for the magnets, with the sintered form exhibiting marginally stronger magnetic properties.
Prior to the Samarium Cobalt magnet, the standard was the Alnico alloy combination magnet, or Aluminum-Nickel-Cobalt. This alloy magnet was used until the 1970s, when rare earth metals began making their way into the magnet industry. Although termed a “rare earth metal” based on its location on the periodic table of elements, neodymium exists plentifully on Earth’s crust. In fact, it is uncommon for a modern electronic device to lack a Neo magnet. The “permanent magnet” property does not mean that it will be magnetized until the end of time. Rather, this phrase is used to describe a piece of metal that is naturally magnetized and loses its magnetism at an extremely slow rate. Once the magnetism is gone, it can not be re-magnetized. On the other hand, a non-permanent magnet can be re-magnetized infinitely, though the magnetic properties will not last for a long duration.
Properties
Neodymium, or neo, magnets are very powerful for their size. Much like ants can lift many times their own weight, a neo magnet can hold a substance with up to 1,300 times its own weight (Fig. 1). The different processes utilized (powder-bonded and sintered) to create the magnet can also result in varying properties, with neither having a distinct advantage over the other although some may argue that the bonded process is better [3]. Some of the properties used to judge the effectiveness of a magnet are remanence, the measure of the strength of the magnetic field; coercivity, the resistance to becoming demagnetized; and the Curie temperature, the temperature at which the magnet becomes demagnetized.
The magnetic field of magnet is measured in the SI unit of Tesla. A higher value indicates a stronger magnet. According to the Magnetic Materials Producers Association (MMPA), a neo magnet exhibits magnetic field strengths of 1.0-1.4 Teslas. The Samarium Cobalt only displays 0.8 to 1.1 Teslas. A common ceramic magnet only exhibits field of 0.5 to 1.0 Teslas. Also unlike its predecessors, neo magnets have less density: 7.3 g/cm3 compared to 8.3 g/cm3 for Samarium Cobalt magnets [4].
Compared to other magnets, neo magnets have higher remanence and coercivity values. However, they have a major drawback of having lower Curie temperatures. The Curie temperature of a neo magnet generally maxes out at about 350°C. While the Curie temperature is lower than that of other magnets, 350°C is more than enough of a threshold for the magnet to be useful for a large majority of household electronics and applications. Conversely, the Samarium Cobalt magnet has a Curie temperature of 800°C, making it more applicable in industry usage [5]. After a magnet has exceeded the Curie temperature, it cannot be re-magnetized and used as a magnet.
Neo magnets are also relatively cheap. A Samarium Cobalt magnet costs approximately $100 per pound, while a neo magnet costs about half of that. A very common ceramic magnet only costs approximately $2 a pound [6]. However, based on the relative cost per BHmax (the density of magnetic energy), the neo magnet comes in at $1.40/BHmax compared to $6.00/BHmax for Samarium Cobalt and $0.50/BHmax for ceramic. Besides being cost-efficient, a neo magnet’s strength also permits its use in a diverse array of applications.
Applications
The strength and permanence of a neo magnet allow it to be used in many electronic devices. Every magnet creates an electromagnetic field that can be used to move a coil and create sound waves [7]. This property allows its use in speakers, headphones, and phones. Neo magnets are also found on hard drives. Overall, since the neo magnet is lighter, stronger, and cheaper than the once-used Samarium Cobalt magnet, it is ideal for making electronics more efficient.
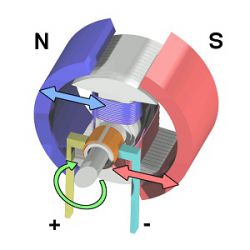
Magnets are also an integral part of an electric motor. An electric motor relies on the concept of “opposites attract and likes repel” to obtain the mechanical energy required to move; two distinct magnets are placed in such a way that the opposite poles of a magnet will keep repelling, allowing one magnet to continuously spin (Fig. 2). This spin creates an electromagnetic field, allowing electrical energy to be converted into mechanical energy. Because the neo magnet is so powerful and permanent, it is an ideal candidate to be used in an electric motor.
The strength and relatively light density of the neo magnets allow for electric motors to be lighter, thus reducing the weight of electric vehicles. Fore example, a hybrid vehicle’s motor, like the Toyota Prius (Fig. 3), houses about 2.2 pounds of neo [8].
Besides being used in electronic devices, neo magnets have many other uses. They can be used as tools around the house, for medicinal or health purposes, in educational applications, or even just for fun. As tools, neo magnets can also be used to find nails in walls, remove metal impurities from car oil, form a magnetic storage rack for various metal items, such as knives or tools [9], or even form the common refrigerator magnet. One example of a medicinal device that utilizes neo magnets is the Magnetic Resonance Imaging (MRI) machine. The magnetic properties of the magnets can also be used for therapy purposes, creating a magnetic flow over the body that can help relieve pain from arthritis [10]. The magnets are also currently marketed as toys, intended to be used in magnetic construction sets, for desktop novelty purposes, or to create sculptures for aesthetic pleasure (Fig. 4).
Recently, the enormous interest in becoming “greener” or more environmentally friendly has resulted in an increase in sales of hybrid vehicles. At the heart of every hybrid vehicle is an electric engine that requires the use of a strong magnet like neodymium. New smart phones also put a strain on the supply of neodymium. Currently, most of the world’s neodymium is mined in China. However, China has recently begun to limit its exports of neodymium, causing concerns of shortages worldwide [11]. Without another large source of neodymium, America will no longer be able to produce the smart phones or hybrid vehicles sold every day. Although there are other mines located domestically and in Canada, the shortage is cause for concern.
With 28 years having elapsed since the discovery of neo magnets, perhaps a new combination of materials can be molded together to create a magnet with properties that rival the neo magnet. If the current demand for electronics continues, the world’s supply of neodymium may be in danger of running out. These shortages and concerns can hopefully bring to light Earth’s limited resources and perhaps attract some attention.